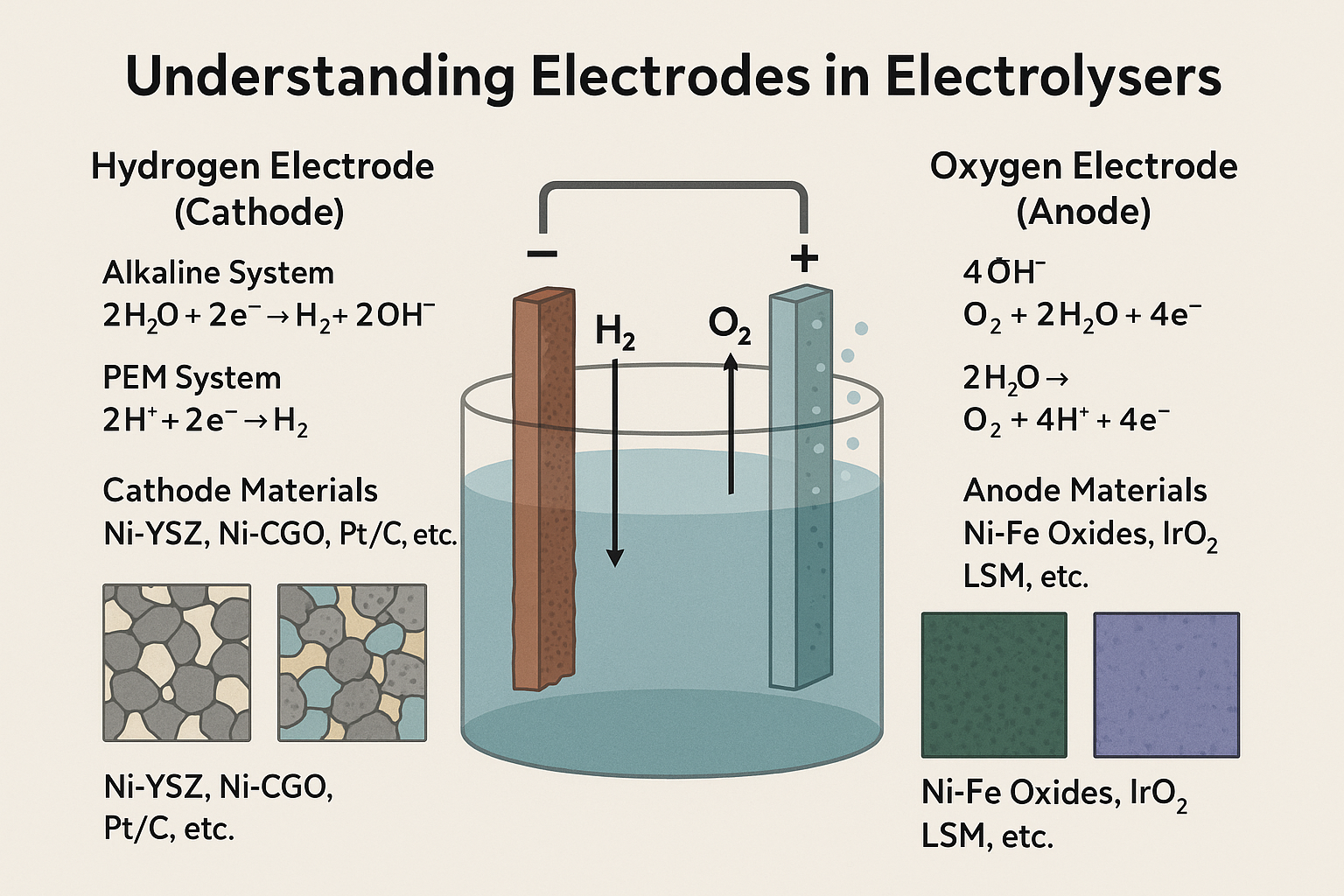
In electrochemical hydrogen production, particularly via water electrolysis, the performance of both the hydrogen electrode (cathode) and oxygen electrode (anode) is fundamental to system efficiency, stability, and cost. Despite receiving limited attention in public discourse, electrodes are among the most critical components influencing the viability of green hydrogen as a scalable energy solution.
1. Electrochemical Functionality
Cathode (Hydrogen Electrode)
At the cathode, the Hydrogen Evolution Reaction (HER) takes place:
- In Alkaline Systems: 2H2O + 2e− → H2 + 2OH−
- In PEM Systems: 2H+ + 2e− → H2
HER occurs at triple phase boundaries (TPBs), the junctions where the electron-conducting phase (e.g., Ni or Pt), the ion-conducting electrolyte, and the gaseous phase (water vapor or hydrogen) meet. The cathode must be porous, electronically conductive, and chemically stable under reducing conditions.
Anode (Oxygen Electrode)
At the anode, the Oxygen Evolution Reaction (OER) takes place:
- In Alkaline Systems: 4OH− → O2 + 2H2O + 4e−
- In PEM Systems: 2H2O → O2 + 4H+ + 4e−
The anode must withstand oxidizing conditions, have excellent electrocatalytic activity for OER, and maintain ionic conductivity while supporting gas evolution.
2. Materials and Structure
Cathode Materials
- Ni-YSZ (Nickel–Yttria Stabilized Zirconia) in SOECs
- Ni-CGO (Nickel–Cerium Gadolinium Oxide) in CGO-based systems
- PEM Systems: Pt-based catalysts supported on carbon or Ti substrates
These materials are engineered to have high porosity, percolation pathways, and mechanical strength under thermal cycling.
Anode Materials
- Alkaline Systems: Ni-Fe, Ni-Co oxides, or perovskites
- PEM Electrolysers: Iridium Oxide (IrO2) or Ruthenium Oxide (RuO2) – SOECs: LSM or LSCF
The anode must balance catalytic efficiency, corrosion resistance, and long-term structural integrity.
3. Performance Metrics
Cathode KPIs
- Overpotential
- Exchange Current Density
- Polarization Resistance
- Degradation Mechanisms (e.g., Ni particle coarsening, delamination)
Anode KPIs
- OER Overpotential
- Stability Under Oxidizing Conditions
- Catalyst Utilization
- Failure Modes (e.g., catalyst dissolution, cracking)
4. Reference Role and Measurement Standard
While the Hydrogen Electrode often serves as a reference in electrochemistry (e.g., Standard Hydrogen Electrode – SHE), the Oxygen Electrode is crucial in benchmarking OER performance
and voltage efficiency in full-cell operation.
- SHE (0 V) forms the baseline for all electrode potentials but is rarely used practically.
- RHE (Reversible Hydrogen Electrode) adjusts for pH and is more widely adopted.
- Practical reference systems like Ag/AgCl and Calomel electrodes are used depending on electrolyte compatibility.
5. Future Development Areas
Cathode R&D Focus
- PGM-Free Catalysts: Ni-Mo, Ni-Fe, transition metal compounds – 3D Architectures
- Carbon-Free Designs
- Dynamic Load Adaptability
Anode R&D Focus
- Reducing PGM Usage
- Hybrid Oxide Catalysts
- Stability Under O2 Evolution – Coating Techniques
Conclusion
The electrodes in an electrolyser, both cathode and anode, are foundational to green hydrogen production. Their material composition, structure, and degradation behaviour directly impact system efficiency, durability, and total cost of ownership (TCO).
As the hydrogen economy scales up, advanced electrode engineering will be as crucial as innovations in power electronics or electrolyser stack integration. Understanding and optimising both the hydrogen and oxygen electrodes is not merely a research objective, it is a strategic necessity for the future of clean energy.